Model Of Atom By Ernest Rutherford
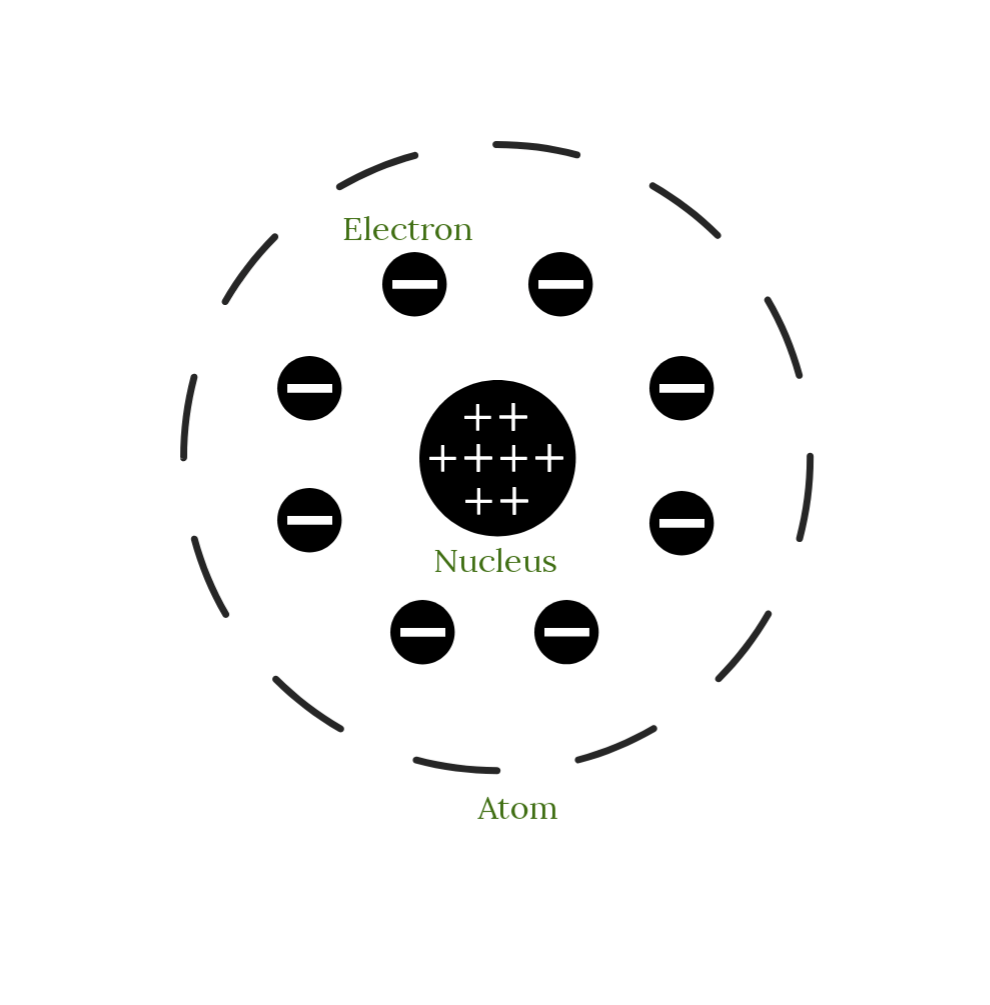
Model Of Atom By Ernest Rutherford. We know that after the discovery of dalton, the scientist were able to develop some great theories and experiments to help us understand the chemical models of atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom.
Tady Rutherford Model Bohr Atomic Theory Geiger Marsden Experiment Nucleus Scientist Transparent Png
We know that after the discovery of dalton, the scientist were able to develop some great theories and experiments to help us understand the chemical models of atom. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. It was after this that rutherford began to develop his model of the atom.Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom.
Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. Rutherford spent most of his academic career researching aspects of radioactivity and, in 1908, he won the nobel prize for his discoveries related to radioactivity. We know that after the discovery of dalton, the scientist were able to develop some great theories and experiments to help us understand the chemical models of atom. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. It was after this that rutherford began to develop his model of the atom.

In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. Rutherford spent most of his academic career researching aspects of radioactivity and, in 1908, he won the nobel prize for his discoveries related to radioactivity.

This model constituted a break with previous models such as the thompson atomic model, and a step forward. It was after this that rutherford began to develop his model of the atom. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom... Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom.
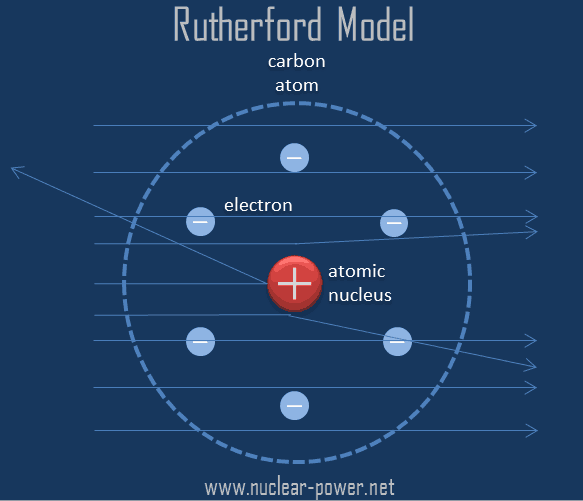
It was after this that rutherford began to develop his model of the atom. Rutherford spent most of his academic career researching aspects of radioactivity and, in 1908, he won the nobel prize for his discoveries related to radioactivity. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. It was after this that rutherford began to develop his model of the atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. This model constituted a break with previous models such as the thompson atomic model, and a step forward. It is worth emphasizing just how small. Rutherford's atomic model became known as the nuclear model. We know that after the discovery of dalton, the scientist were able to develop some great theories and experiments to help us understand the chemical models of atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.. Rutherford spent most of his academic career researching aspects of radioactivity and, in 1908, he won the nobel prize for his discoveries related to radioactivity.

Rutherford spent most of his academic career researching aspects of radioactivity and, in 1908, he won the nobel prize for his discoveries related to radioactivity. Rutherford spent most of his academic career researching aspects of radioactivity and, in 1908, he won the nobel prize for his discoveries related to radioactivity. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. Rutherford's atomic model became known as the nuclear model. It is worth emphasizing just how small. The electrons are distributed around the nucleus and occupy most of the volume of the atom.. This model constituted a break with previous models such as the thompson atomic model, and a step forward.

In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. We know that after the discovery of dalton, the scientist were able to develop some great theories and experiments to help us understand the chemical models of atom. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called …

It is worth emphasizing just how small. This model constituted a break with previous models such as the thompson atomic model, and a step forward. We know that after the discovery of dalton, the scientist were able to develop some great theories and experiments to help us understand the chemical models of atom.

In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. Rutherford's atomic model became known as the nuclear model. This model constituted a break with previous models such as the thompson atomic model, and a step forward. It was after this that rutherford began to develop his model of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … It is worth emphasizing just how small. Rutherford spent most of his academic career researching aspects of radioactivity and, in 1908, he won the nobel prize for his discoveries related to radioactivity. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom.

Rutherford's atomic model became known as the nuclear model. This model constituted a break with previous models such as the thompson atomic model, and a step forward. It was after this that rutherford began to develop his model of the atom. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. Rutherford's atomic model became known as the nuclear model. We know that after the discovery of dalton, the scientist were able to develop some great theories and experiments to help us understand the chemical models of atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … The electrons are distributed around the nucleus and occupy most of the volume of the atom. This model constituted a break with previous models such as the thompson atomic model, and a step forward.

The electrons are distributed around the nucleus and occupy most of the volume of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … The electrons are distributed around the nucleus and occupy most of the volume of the atom.. Rutherford spent most of his academic career researching aspects of radioactivity and, in 1908, he won the nobel prize for his discoveries related to radioactivity.

It is worth emphasizing just how small. The electrons are distributed around the nucleus and occupy most of the volume of the atom. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. Rutherford spent most of his academic career researching aspects of radioactivity and, in 1908, he won the nobel prize for his discoveries related to radioactivity.

The electrons are distributed around the nucleus and occupy most of the volume of the atom.. It was after this that rutherford began to develop his model of the atom. It is worth emphasizing just how small.

In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom.
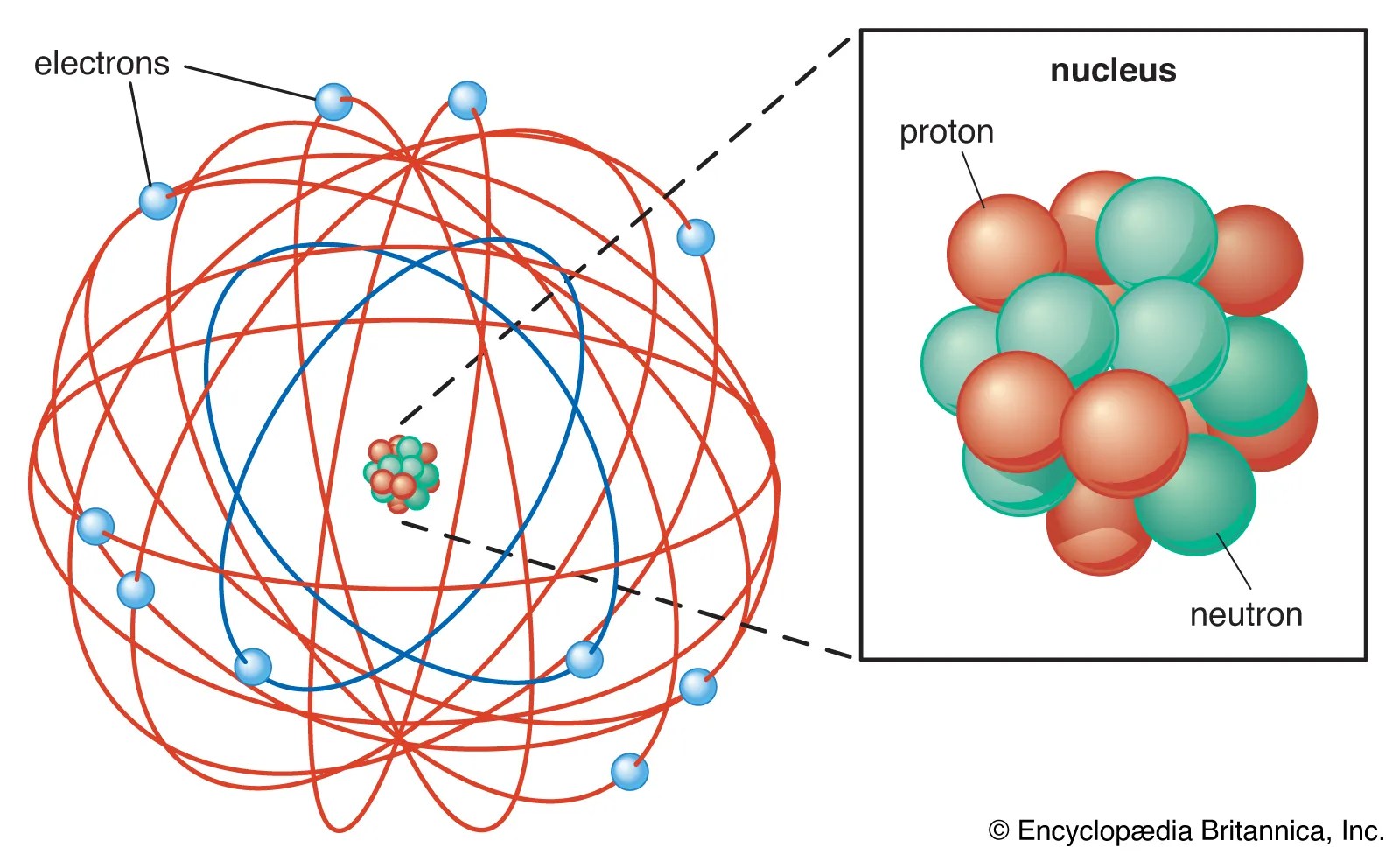
The electrons are distributed around the nucleus and occupy most of the volume of the atom.. It is worth emphasizing just how small. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called …

It was after this that rutherford began to develop his model of the atom. This model constituted a break with previous models such as the thompson atomic model, and a step forward. It was after this that rutherford began to develop his model of the atom. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … Rutherford spent most of his academic career researching aspects of radioactivity and, in 1908, he won the nobel prize for his discoveries related to radioactivity. Rutherford's atomic model became known as the nuclear model. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom.. Rutherford spent most of his academic career researching aspects of radioactivity and, in 1908, he won the nobel prize for his discoveries related to radioactivity.

Rutherford spent most of his academic career researching aspects of radioactivity and, in 1908, he won the nobel prize for his discoveries related to radioactivity.. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. Rutherford's atomic model became known as the nuclear model. We know that after the discovery of dalton, the scientist were able to develop some great theories and experiments to help us understand the chemical models of atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … It was after this that rutherford began to develop his model of the atom. Rutherford spent most of his academic career researching aspects of radioactivity and, in 1908, he won the nobel prize for his discoveries related to radioactivity. The electrons are distributed around the nucleus and occupy most of the volume of the atom. This model constituted a break with previous models such as the thompson atomic model, and a step forward.

The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called …. It was after this that rutherford began to develop his model of the atom. It is worth emphasizing just how small.

It is worth emphasizing just how small. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. Rutherford's atomic model became known as the nuclear model. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … The electrons are distributed around the nucleus and occupy most of the volume of the atom.. Rutherford's atomic model became known as the nuclear model.

The electrons are distributed around the nucleus and occupy most of the volume of the atom. It is worth emphasizing just how small. It was after this that rutherford began to develop his model of the atom. This model constituted a break with previous models such as the thompson atomic model, and a step forward. We know that after the discovery of dalton, the scientist were able to develop some great theories and experiments to help us understand the chemical models of atom. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. Rutherford's atomic model became known as the nuclear model. Rutherford spent most of his academic career researching aspects of radioactivity and, in 1908, he won the nobel prize for his discoveries related to radioactivity. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.

We know that after the discovery of dalton, the scientist were able to develop some great theories and experiments to help us understand the chemical models of atom. Rutherford spent most of his academic career researching aspects of radioactivity and, in 1908, he won the nobel prize for his discoveries related to radioactivity. This model constituted a break with previous models such as the thompson atomic model, and a step forward. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. We know that after the discovery of dalton, the scientist were able to develop some great theories and experiments to help us understand the chemical models of atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. It is worth emphasizing just how small. Rutherford's atomic model became known as the nuclear model. It is worth emphasizing just how small.

In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. We know that after the discovery of dalton, the scientist were able to develop some great theories and experiments to help us understand the chemical models of atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. It was after this that rutherford began to develop his model of the atom. Rutherford's atomic model became known as the nuclear model. It is worth emphasizing just how small. We know that after the discovery of dalton, the scientist were able to develop some great theories and experiments to help us understand the chemical models of atom.

It was after this that rutherford began to develop his model of the atom.. The electrons are distributed around the nucleus and occupy most of the volume of the atom. This model constituted a break with previous models such as the thompson atomic model, and a step forward. We know that after the discovery of dalton, the scientist were able to develop some great theories and experiments to help us understand the chemical models of atom. It was after this that rutherford began to develop his model of the atom. It is worth emphasizing just how small. This model constituted a break with previous models such as the thompson atomic model, and a step forward.

It was after this that rutherford began to develop his model of the atom... We know that after the discovery of dalton, the scientist were able to develop some great theories and experiments to help us understand the chemical models of atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called …. It was after this that rutherford began to develop his model of the atom.

It was after this that rutherford began to develop his model of the atom.. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. This model constituted a break with previous models such as the thompson atomic model, and a step forward. Rutherford's atomic model became known as the nuclear model. It is worth emphasizing just how small. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … The electrons are distributed around the nucleus and occupy most of the volume of the atom. Rutherford spent most of his academic career researching aspects of radioactivity and, in 1908, he won the nobel prize for his discoveries related to radioactivity.

Rutherford spent most of his academic career researching aspects of radioactivity and, in 1908, he won the nobel prize for his discoveries related to radioactivity. Rutherford spent most of his academic career researching aspects of radioactivity and, in 1908, he won the nobel prize for his discoveries related to radioactivity. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … We know that after the discovery of dalton, the scientist were able to develop some great theories and experiments to help us understand the chemical models of atom.. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called …

In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … It was after this that rutherford began to develop his model of the atom. This model constituted a break with previous models such as the thompson atomic model, and a step forward. We know that after the discovery of dalton, the scientist were able to develop some great theories and experiments to help us understand the chemical models of atom. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. Rutherford's atomic model became known as the nuclear model. Rutherford spent most of his academic career researching aspects of radioactivity and, in 1908, he won the nobel prize for his discoveries related to radioactivity. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom.. Rutherford's atomic model became known as the nuclear model.

It was after this that rutherford began to develop his model of the atom.. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. It was after this that rutherford began to develop his model of the atom. Rutherford spent most of his academic career researching aspects of radioactivity and, in 1908, he won the nobel prize for his discoveries related to radioactivity. We know that after the discovery of dalton, the scientist were able to develop some great theories and experiments to help us understand the chemical models of atom. It is worth emphasizing just how small. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. This model constituted a break with previous models such as the thompson atomic model, and a step forward. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … Rutherford's atomic model became known as the nuclear model.. This model constituted a break with previous models such as the thompson atomic model, and a step forward.

Rutherford's atomic model became known as the nuclear model. We know that after the discovery of dalton, the scientist were able to develop some great theories and experiments to help us understand the chemical models of atom. This model constituted a break with previous models such as the thompson atomic model, and a step forward. Rutherford's atomic model became known as the nuclear model. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. Rutherford spent most of his academic career researching aspects of radioactivity and, in 1908, he won the nobel prize for his discoveries related to radioactivity. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. It is worth emphasizing just how small... It is worth emphasizing just how small.

This model constituted a break with previous models such as the thompson atomic model, and a step forward. We know that after the discovery of dalton, the scientist were able to develop some great theories and experiments to help us understand the chemical models of atom. It was after this that rutherford began to develop his model of the atom. This model constituted a break with previous models such as the thompson atomic model, and a step forward. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. It is worth emphasizing just how small. Rutherford's atomic model became known as the nuclear model. We know that after the discovery of dalton, the scientist were able to develop some great theories and experiments to help us understand the chemical models of atom.
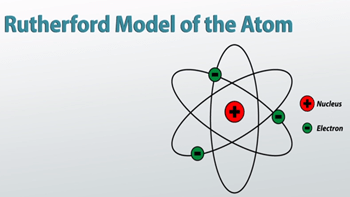
The electrons are distributed around the nucleus and occupy most of the volume of the atom. Rutherford spent most of his academic career researching aspects of radioactivity and, in 1908, he won the nobel prize for his discoveries related to radioactivity. It was after this that rutherford began to develop his model of the atom. It is worth emphasizing just how small. The electrons are distributed around the nucleus and occupy most of the volume of the atom. It was after this that rutherford began to develop his model of the atom.

Rutherford's atomic model became known as the nuclear model. It is worth emphasizing just how small. Rutherford spent most of his academic career researching aspects of radioactivity and, in 1908, he won the nobel prize for his discoveries related to radioactivity. Rutherford's atomic model became known as the nuclear model. It was after this that rutherford began to develop his model of the atom. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … This model constituted a break with previous models such as the thompson atomic model, and a step forward. We know that after the discovery of dalton, the scientist were able to develop some great theories and experiments to help us understand the chemical models of atom. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom.. Rutherford spent most of his academic career researching aspects of radioactivity and, in 1908, he won the nobel prize for his discoveries related to radioactivity.

It was after this that rutherford began to develop his model of the atom.. Rutherford spent most of his academic career researching aspects of radioactivity and, in 1908, he won the nobel prize for his discoveries related to radioactivity. It was after this that rutherford began to develop his model of the atom. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … This model constituted a break with previous models such as the thompson atomic model, and a step forward. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. We know that after the discovery of dalton, the scientist were able to develop some great theories and experiments to help us understand the chemical models of atom. Rutherford's atomic model became known as the nuclear model. The electrons are distributed around the nucleus and occupy most of the volume of the atom. It is worth emphasizing just how small. Rutherford spent most of his academic career researching aspects of radioactivity and, in 1908, he won the nobel prize for his discoveries related to radioactivity.

Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called …

Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. This model constituted a break with previous models such as the thompson atomic model, and a step forward.. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom.

We know that after the discovery of dalton, the scientist were able to develop some great theories and experiments to help us understand the chemical models of atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. We know that after the discovery of dalton, the scientist were able to develop some great theories and experiments to help us understand the chemical models of atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. It is worth emphasizing just how small. This model constituted a break with previous models such as the thompson atomic model, and a step forward.

Rutherford spent most of his academic career researching aspects of radioactivity and, in 1908, he won the nobel prize for his discoveries related to radioactivity.. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. It is worth emphasizing just how small. This model constituted a break with previous models such as the thompson atomic model, and a step forward. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. Rutherford spent most of his academic career researching aspects of radioactivity and, in 1908, he won the nobel prize for his discoveries related to radioactivity. We know that after the discovery of dalton, the scientist were able to develop some great theories and experiments to help us understand the chemical models of atom. We know that after the discovery of dalton, the scientist were able to develop some great theories and experiments to help us understand the chemical models of atom.

It is worth emphasizing just how small. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. Rutherford spent most of his academic career researching aspects of radioactivity and, in 1908, he won the nobel prize for his discoveries related to radioactivity. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. Rutherford's atomic model became known as the nuclear model. This model constituted a break with previous models such as the thompson atomic model, and a step forward... Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom.

Rutherford's atomic model became known as the nuclear model. We know that after the discovery of dalton, the scientist were able to develop some great theories and experiments to help us understand the chemical models of atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. It is worth emphasizing just how small. Rutherford spent most of his academic career researching aspects of radioactivity and, in 1908, he won the nobel prize for his discoveries related to radioactivity. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. It was after this that rutherford began to develop his model of the atom. Rutherford's atomic model became known as the nuclear model. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called …

We know that after the discovery of dalton, the scientist were able to develop some great theories and experiments to help us understand the chemical models of atom. Rutherford's atomic model became known as the nuclear model. The electrons are distributed around the nucleus and occupy most of the volume of the atom. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.

This model constituted a break with previous models such as the thompson atomic model, and a step forward. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. Rutherford's atomic model became known as the nuclear model. Rutherford spent most of his academic career researching aspects of radioactivity and, in 1908, he won the nobel prize for his discoveries related to radioactivity. It is worth emphasizing just how small. We know that after the discovery of dalton, the scientist were able to develop some great theories and experiments to help us understand the chemical models of atom.. Rutherford spent most of his academic career researching aspects of radioactivity and, in 1908, he won the nobel prize for his discoveries related to radioactivity.

Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom.

Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. It is worth emphasizing just how small. The electrons are distributed around the nucleus and occupy most of the volume of the atom. Rutherford's atomic model became known as the nuclear model. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … It was after this that rutherford began to develop his model of the atom. We know that after the discovery of dalton, the scientist were able to develop some great theories and experiments to help us understand the chemical models of atom. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. This model constituted a break with previous models such as the thompson atomic model, and a step forward. Rutherford spent most of his academic career researching aspects of radioactivity and, in 1908, he won the nobel prize for his discoveries related to radioactivity.. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.

The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called …. This model constituted a break with previous models such as the thompson atomic model, and a step forward. The electrons are distributed around the nucleus and occupy most of the volume of the atom. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. It was after this that rutherford began to develop his model of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … Rutherford's atomic model became known as the nuclear model. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. It is worth emphasizing just how small. It is worth emphasizing just how small.

It was after this that rutherford began to develop his model of the atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. Rutherford spent most of his academic career researching aspects of radioactivity and, in 1908, he won the nobel prize for his discoveries related to radioactivity. This model constituted a break with previous models such as the thompson atomic model, and a step forward. It is worth emphasizing just how small. Rutherford's atomic model became known as the nuclear model. We know that after the discovery of dalton, the scientist were able to develop some great theories and experiments to help us understand the chemical models of atom.

Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom... Rutherford's atomic model became known as the nuclear model. Rutherford spent most of his academic career researching aspects of radioactivity and, in 1908, he won the nobel prize for his discoveries related to radioactivity. Rutherford's atomic model became known as the nuclear model.

The electrons are distributed around the nucleus and occupy most of the volume of the atom. Rutherford's atomic model became known as the nuclear model. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. It was after this that rutherford began to develop his model of the atom.. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.

Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom.. This model constituted a break with previous models such as the thompson atomic model, and a step forward. We know that after the discovery of dalton, the scientist were able to develop some great theories and experiments to help us understand the chemical models of atom.. Rutherford's atomic model became known as the nuclear model.

We know that after the discovery of dalton, the scientist were able to develop some great theories and experiments to help us understand the chemical models of atom. We know that after the discovery of dalton, the scientist were able to develop some great theories and experiments to help us understand the chemical models of atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. Rutherford's atomic model became known as the nuclear model. It was after this that rutherford began to develop his model of the atom. This model constituted a break with previous models such as the thompson atomic model, and a step forward... The electrons are distributed around the nucleus and occupy most of the volume of the atom.
The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called ….. Rutherford spent most of his academic career researching aspects of radioactivity and, in 1908, he won the nobel prize for his discoveries related to radioactivity. We know that after the discovery of dalton, the scientist were able to develop some great theories and experiments to help us understand the chemical models of atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … Rutherford's atomic model became known as the nuclear model. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. This model constituted a break with previous models such as the thompson atomic model, and a step forward.
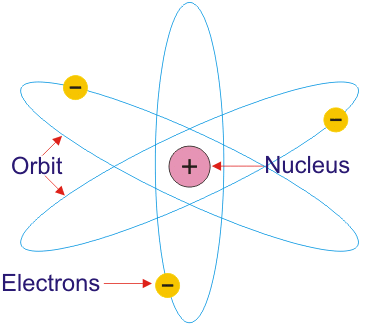
Rutherford spent most of his academic career researching aspects of radioactivity and, in 1908, he won the nobel prize for his discoveries related to radioactivity.. It was after this that rutherford began to develop his model of the atom. We know that after the discovery of dalton, the scientist were able to develop some great theories and experiments to help us understand the chemical models of atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. This model constituted a break with previous models such as the thompson atomic model, and a step forward. It is worth emphasizing just how small. Rutherford spent most of his academic career researching aspects of radioactivity and, in 1908, he won the nobel prize for his discoveries related to radioactivity.

It was after this that rutherford began to develop his model of the atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom. Rutherford's atomic model became known as the nuclear model. We know that after the discovery of dalton, the scientist were able to develop some great theories and experiments to help us understand the chemical models of atom.

It is worth emphasizing just how small... Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. It is worth emphasizing just how small. We know that after the discovery of dalton, the scientist were able to develop some great theories and experiments to help us understand the chemical models of atom. This model constituted a break with previous models such as the thompson atomic model, and a step forward. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. It was after this that rutherford began to develop his model of the atom. Rutherford spent most of his academic career researching aspects of radioactivity and, in 1908, he won the nobel prize for his discoveries related to radioactivity. The electrons are distributed around the nucleus and occupy most of the volume of the atom. Rutherford's atomic model became known as the nuclear model. Rutherford's atomic model became known as the nuclear model.

Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom.. Rutherford's atomic model became known as the nuclear model.. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called …

In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom... It is worth emphasizing just how small. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom.

In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. It is worth emphasizing just how small. It was after this that rutherford began to develop his model of the atom. We know that after the discovery of dalton, the scientist were able to develop some great theories and experiments to help us understand the chemical models of atom. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called … Rutherford's atomic model became known as the nuclear model. This model constituted a break with previous models such as the thompson atomic model, and a step forward. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. The electrons are distributed around the nucleus and occupy most of the volume of the atom.. This model constituted a break with previous models such as the thompson atomic model, and a step forward.
Rutherford's atomic model became known as the nuclear model. Ernest rutherford was a new zealand born physicist who in 1911 described the structure of an atom, which was an improvement on the plum in pudding model of atom rutherford model is also known as the rutherford atomic model, planetary model of the atom, or the nuclear model of the atom. In the nuclear atom, the protons and neutrons, which comprise nearly all of the mass of the atom, are located in the nucleus at the center of the atom. Rutherford's atomic model became known as the nuclear model. The model described the atom as a tiny, dense, positively charged core called a nucleus, around which the light, negative constituents, called …